Challenges facing the distribution of COVID-19 vaccine in Canada, the U.S. and Mexico
The distribution of COVID-19 vaccines has started in North America. What challenges will the carriers that work in the supply chain face?
It’s been less than a year since scientists deciphered the genome of SARS-CoV-2, the virus that causes COVID-19, and pharmaceutical companies have developed the vaccine against it in record time. However, the vaccination process has just begun.
The design of these supply chains is the most complicated that we have ever faced. It must comply with all the technical requirements so that all stages of the transportation, storage, distribution and vaccination, can be performed within a consistent cold chain.
Success requires logistic experts and several special units for aerial and terrestrial transportation. Besides, everything must be secured and marked with serial numbers, codes, and other special anti-counterfeiting systems for traceability.

First challenge: ultracold
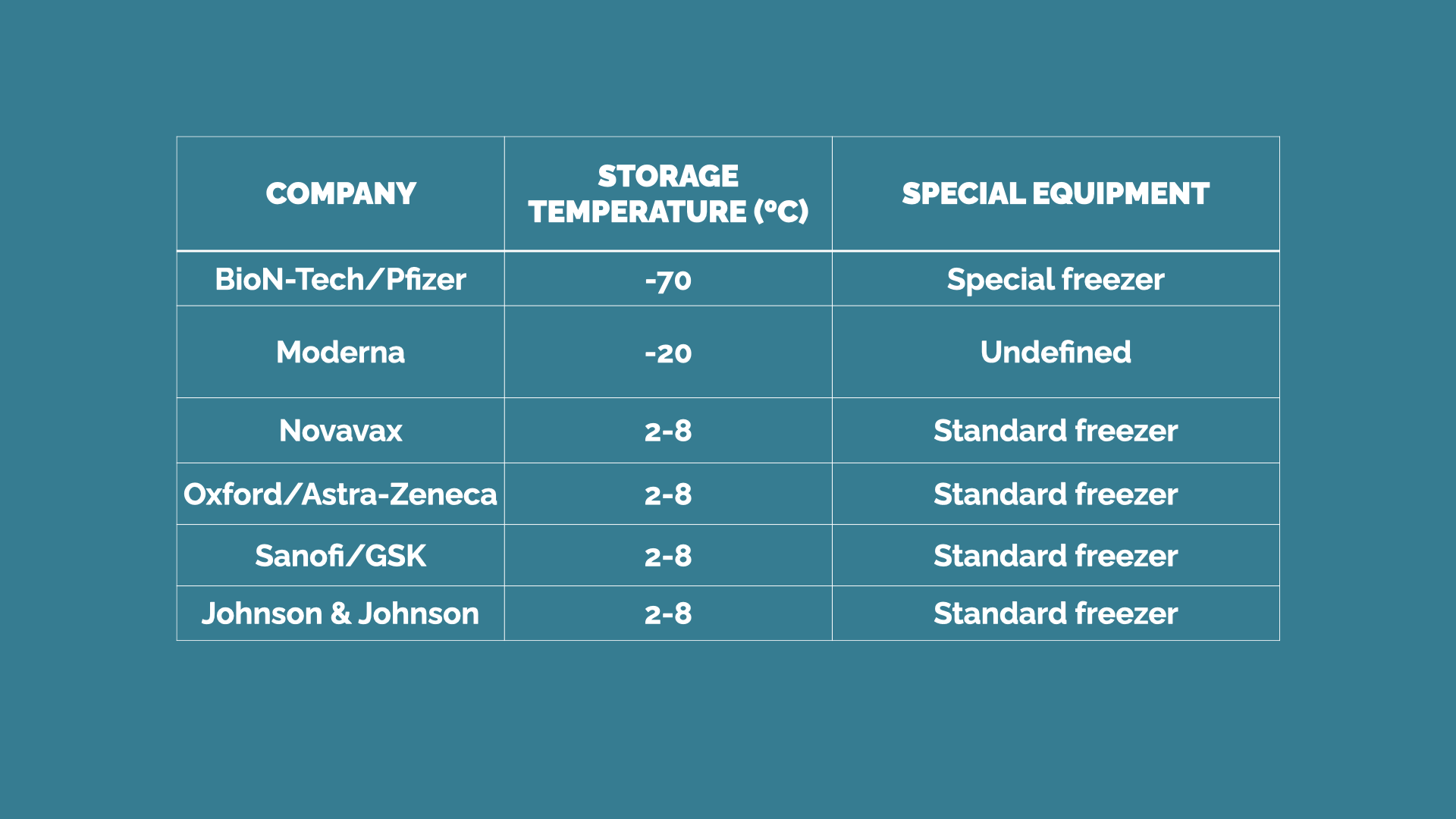
The Phizer-BioNTech vaccine offers 95% protection and works with synthetic ribonucleic acid (RNA) to activate the immune system against the virus. However, this is the vaccine that represents the harder logistic challenge, since it must be carried in a special ultracold freezer at -70ºC, even colder than the Antarctic. This characteristic will make transportation even harder in places where the weather is warm and distances without the required infrastructure are longer.
On the other hand, the Moderna vaccine must be stored at a temperature of -20ºC, which is the standard for most commercial freezers. The rest of the vaccines in the last stages of their development are not formulated with the ARN so they will be easier to work with.
Second challenge: warehousing and traceability
Reception and warehousing locations are the most important aspects right now, then we can develop strategies to take vaccines up to vaccination centers. When we get there, there will be new challenges ahead, since hospitals, medical facilities, and clinics need to have enough staff, equipment, and room to apply the vaccine.
In the first few months, there is a chance that companies and governments use direct store delivery, from the plant to the final destination by aerial or terrestrial transportation, which makes sense in the first part of the process and when vaccination centers are located near the manufacturing site.
Later on, long term storage must be avoided through cross-docking, since this will optimize deliveries to the final destination, through a capillary distribution system, and will reduce the need for warehouses.
The use of local distribution centers has been considered as a possibility to guarantee stocks, however, the execution of this plan will require larger warehouses with appropriate infrastructure for conservation and storage of a large number of vaccines.
It is a fact that all logistic models will be adjusted on the go.
Third challenge: physical and cybernetic security
Just as we said, taking care of vaccine batches and to tag them with serial codes will be the first step to bulletproof the supply chain. The pharmaceutical industry has the technology it needs, but it never had to move several vaccines this large in so little time before.
The distribution center must keep a First Expired, First Out (FEFO) system during shipping. At this point, the most important thing is to establish distribution places and delivery date priorities.
When we talk about security, we do not mean the physical one only. During the first days of December, cyber-attacks to the cold chain in six countries were reported. They used phishing through emails sent to the Cold Chain Equipment Optimization Platform (CCEOP) of Gavi, the international Vaccine Alliance.
According to the IBM statement, attackers made themselves seem like representatives of the Chinese company Haier Biomedical. The email contained an HTML that demanded passwords of the receivers, which could have been used for the wrong purpose.
The attack was aimed to organizations involved in the manufacture of solar panels, used to keep vaccines cold in places without electricity; a development company in South Korea and a German website developer whose clients are pharmaceutical manufacturers, container transportation carriers, biotechnology and the production of electric components used in communication systems. What this means is that all the ones involved in the vaccine supply chain must bulletproof their processes without further delay.

Impact of the COVID-19 vaccine distribution on freight transportation
Shippers that are not involved in the reefer business are worried about not being a priority and that rates might suffer a rapid increase so high that it would prevent them from making their shipments.
On the other hand, shippers that have reefers and refrigerated warehouses and have experience with pharmaceutical loads do not know if they could work with vaccine shipments. One of their worries is that their storage capacity could be not large enough even if they get the call to help with the emergency.
In regards to 3PL, they should adapt and work on doing the paperwork to obtain the credentials they need to work with pharmaceutical loads.
As it has happened during the whole pandemic, links in the supply chain will obtain more experience and reliability while the foreign and local shipments go on.
Mexicom Logistics work to keep supply chains in motion without disruptions through North America in the COVID-19 pandemic; besides, we offer southbound reefers from Canada and the U.S. to Mexico, as well as cold transshipment in Laredo.
Sources:
https://globalnews.ca/news/7535034/coronavirus-vaccine-moderna-approval-weeks-health-canada/
https://coronavirus.gob.mx/wp-content/uploads/2020/12/PolVx_COVID.pdf
http://t21.com.mx/logistica/2020/12/04/desafio-logistico-vacuna-contra-covid-19
https://www.theverge.com/2020/12/3/22151016/hackers-phishing-coronavirus-vaccine-ibm-security









